"Prevent Breast Cancer."
Simple DNA Saliva Test To Uncover Your Inherited Risk For Breast Cancer
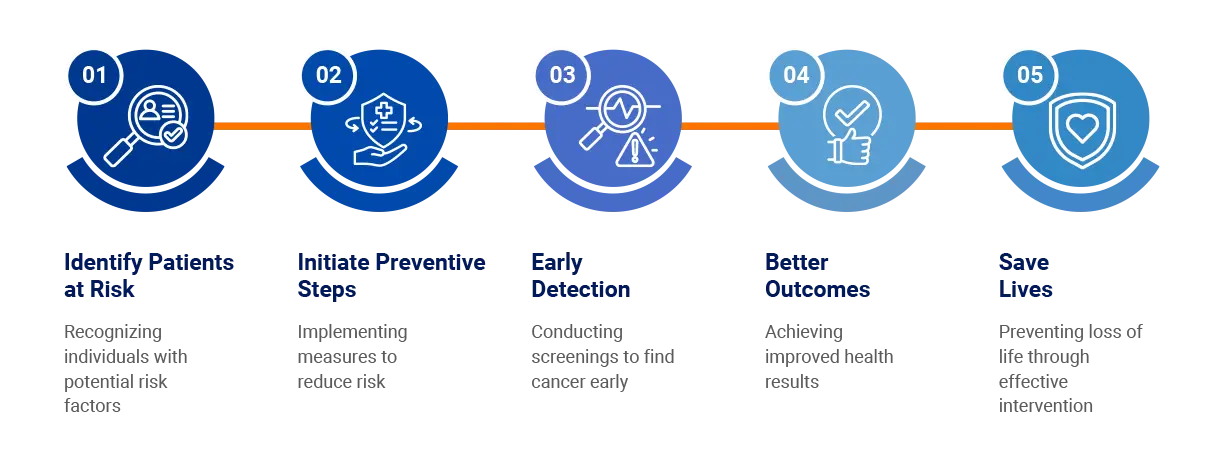
Breast Cancer Risk Management Sequence
What Is A Polygenic Score?
- Everyone has small genetic differences, called variants.
- These variants can affect the risk of developing certain diseases.
- A polygenic risk score (PRS) combines the effects of millions of genetic variants to estimate your disease risk.
- While each variant has a small effect, their combined impact can be significant.


Who Should Take This Test?
Female – Adult (18-75 years)
Healthy women interested in understanding their personal risk of breast cancer.
Women diagnosed with breast cancer seeking to understand the genetic factors contributing to their disease.
Women who carry a rare monogenic mutation (e.g., BRCA) associated with increased breast cancer risk.
Women with a family history of breast cancer.
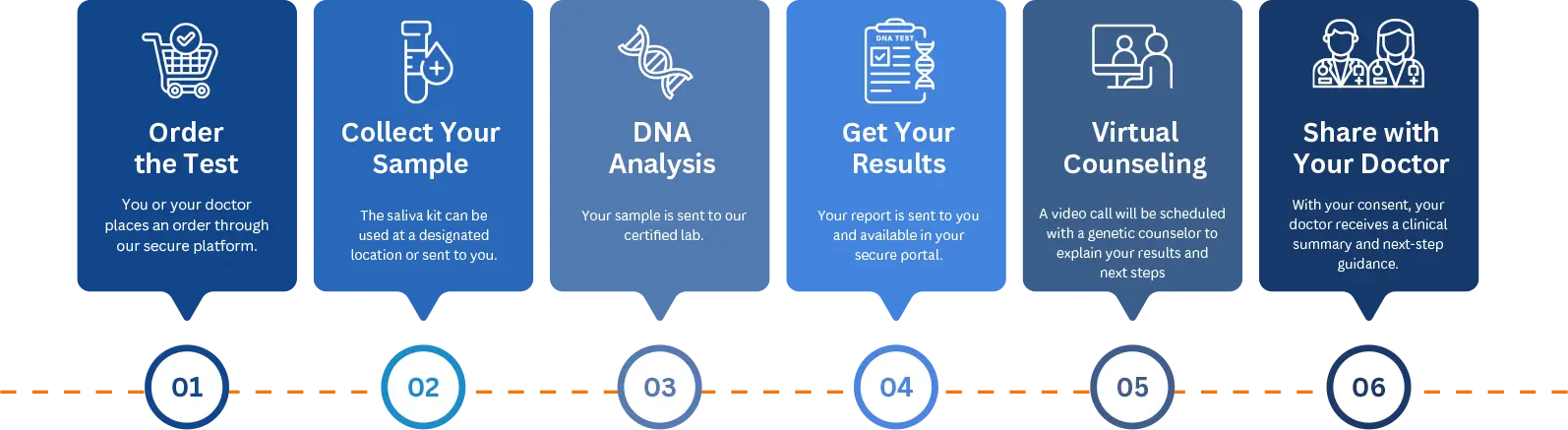
How It Works - Step By Step
The test is powered by the innovative Blended Genome Exome™ technology, developed in collaboration with Broad Clinical Labs at Harvard University and the Massachusetts Institute of Technology.
The technology combines 95.5× whole-exome sequencing (WES) with genome-wide low-coverage whole-genome sequencing (lcWGS) to detect both rare pathogenic variants and polygenic risk with high analytical precision.
Testing is performed through CAP/CLIA accredited laboratories, with advanced analytics support from Allelica and the Laboratory for Molecular Medicine (Mass General Brigham), and regional collaboration with Novo Genomics.
Equipped with one of the most precise and comprehensive genomic risk assessments available, the test integrates monogenic and polygenic risk into a single, clinically actionable report.
Results are ready in 4 to 8 weeks, enabling you to act early and make informed, proactive health decisions.