"Prevent Prostate Cancer."
Simple DNA Saliva To Uncover Your Inherited Risk For Prostate Cancer
What Is A Polygenic Score?
- Everyone has small genetic differences, called variants.
- These variants can affect the risk of developing certain diseases.
- A polygenic risk score (PRS) combines the effects of millions of genetic variants to estimate your disease risk.
- While each variant has a small effect, their combined impact can be significant.

Who Should Tale This Test?
Healthy Men aged 40 and above interested in understanding their genetic risk
Men with a family history of prostate cancer (especially first-degree relatives)
Men of African or Middle Eastern ancestry, who are at higher genetic risk and underrepresented in traditional screening tools
Men with known monogenic mutations (e.g., BRCA1/2, HOXB13) seeking a more comprehensive risk assessment
Men with elevated PSA levels or inconclusive biopsy results, as part of a broader risk evaluation
Men at average risk considering when and how often to begin screening
Why PRS Matters For Prostate Cancer
Personalized Risk: PRS provides an individualized estimate of lifetime prostate cancer risk based on common genetic variants.
Beyond Family History: It identifies risk in men without a known family history or monogenic mutation.
Stronger Stratification: PRS complements PSA and family history, helping distinguish between low-, intermediate-, and high-risk individuals.
Early Detection: Guides earlier and more frequent screening in high-risk individuals, potentially enabling timely diagnosis and treatment.
Precision Prevention: Informs lifestyle counseling and preventive strategies tailored to genetic risk.
Better Resource Use: Helps reduce over-screening in low-risk men and focus healthcare resources on those most at risk.
How It Works - Step By Step
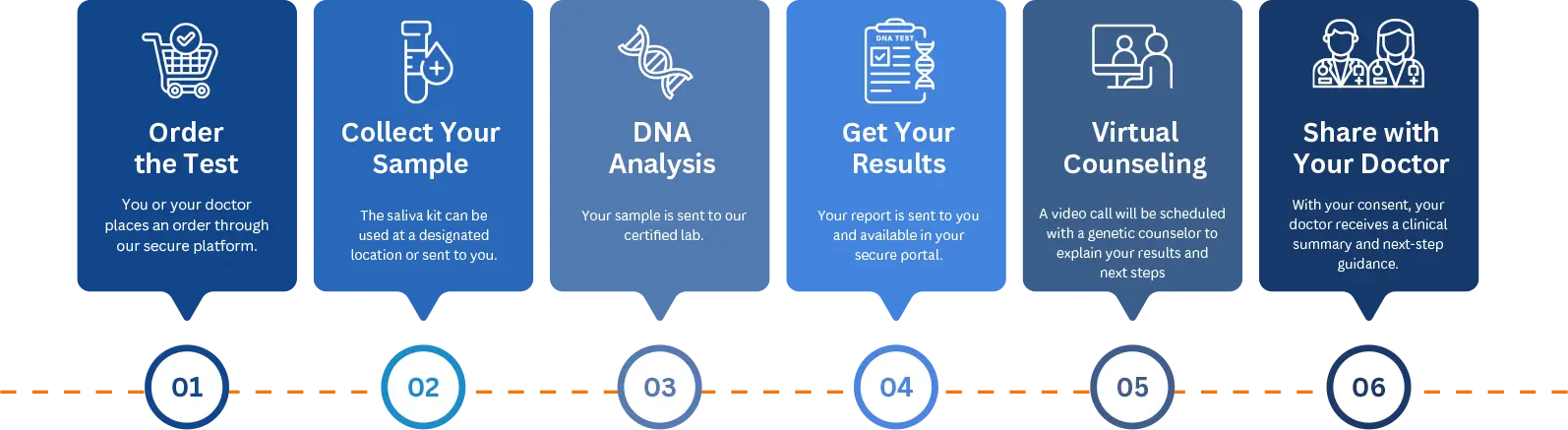
The test is powered by the innovative Blended Genome Exome™ technology, developed in collaboration with Broad Clinical Labs at Harvard University and the Massachusetts Institute of Technology.
The technology combines 95.5× whole-exome sequencing (WES) with genome-wide low-coverage whole-genome sequencing (lcWGS) to detect both rare pathogenic variants and polygenic risk with high analytical precision.
Testing is performed through CAP/CLIA accredited laboratories, with advanced analytics support from Allelica and the Laboratory for Molecular Medicine (Mass General Brigham), and regional collaboration with Novo Genomics.
Equipped with one of the most precise and comprehensive genomic risk assessments available, the test integrates monogenic and polygenic risk into a single, clinically actionable report.
Results are ready in 4 to 8 weeks, enabling you to act early and make informed, proactive health decisions.